The National Institute on Aging (NIA) IMbedded Pragmatic Alzheimer’s disease (AD) and AD-Related Dementias (AD/ADRD) Clinical Trials (IMPACT) Collaboratory has released a new funding opportunity available for investigators interested in developing embedded pragmatic clinical trials (ePCTs) in dementia research.
Detailed information about the program as well as timelines, eligibility, and other requirements are provided below and in the request for applications (RFAs). Updates will be posted to IMPACT Pilot Grant Program webpage.
Pilot Grant Program RFA Cycle 2B
The IMPACT Collaboratory is soliciting letters of intent for several 1-year pilot awards for up to $175,000 in direct costs. The goal of these awards is to support pilot studies to generate the preliminary data necessary to design and conduct future full-scale ePCTs of non-pharmacologic interventions in health care systems for people living with AD/ADRD and/or their care partners. The mandatory letter of intent is due September 18, 2020.
The IMPACT Collaboratory will consider applications for pilot ePCTs testing non-pharmacological interventions in people living with AD/ADRD and their care partners. In this grant cycle, the IMPACT Collaboratory will consider all applications that propose pilot ePCTs for people living with AD/ADRD and their care partners but will prioritize those in the following areas:
- Interventions that strive to reduce inequities in health care experienced by minority, underserved, or disadvantaged populations or in settings serving a disproportionate share of minority, underserved, or disadvantaged populations;
- Interventions that strive to address social isolation and loneliness; and
- Interventions that use telemedicine, telehealth, and remote technologies to improve health, unmet needs, quality of life and/or health outcomes.
The IMPACT Collaboratory will fund up to five 1-year, non-renewable pilot grant awards for up to $175,000 in direct costs. Letters of intent are required and due September 18, 2020. Full proposal applications are by invitation only and will be due January 8, 2021. For further details, please see the on the IMPACT Pilot Grant Program webpage.
Two optional informational webinars will provide investigators with an overview of application details and support that the IMPACT Collaboratory can provide to assist with proposal development (e.g., trial design, measurement, data extraction, etc.). Investigators will have the opportunity to ask questions. Pre-registration is required using the links below.
- Wednesday, August 26, 2020 at 2:00 pm ET: Registration closed.
- Tuesday, September 8, 2020 at 4:00 pm ET: Registration closed.
Registrants will receive a confirmation email containing information to join the webinar.
The IMPACT Collaboratory encourages all eligible researchers to take advantage of this important funding opportunity, which has the potential to advance care for people living with dementia and/or their care partners in real-world settings. For more information about the IMPACT Collaboratory, please visit impactcollaboratory.org.
The NIA IMPACT Collaboratory is supported through funding from the National Institute on Aging (U54AG063546).








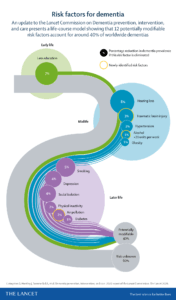 The Lancet Commission on dementia prevention, intervention, and care published a report July 30, 2020 that highlights recommendations for policy makers and individuals to help reduce dementia risk worldwide.
The Lancet Commission on dementia prevention, intervention, and care published a report July 30, 2020 that highlights recommendations for policy makers and individuals to help reduce dementia risk worldwide.