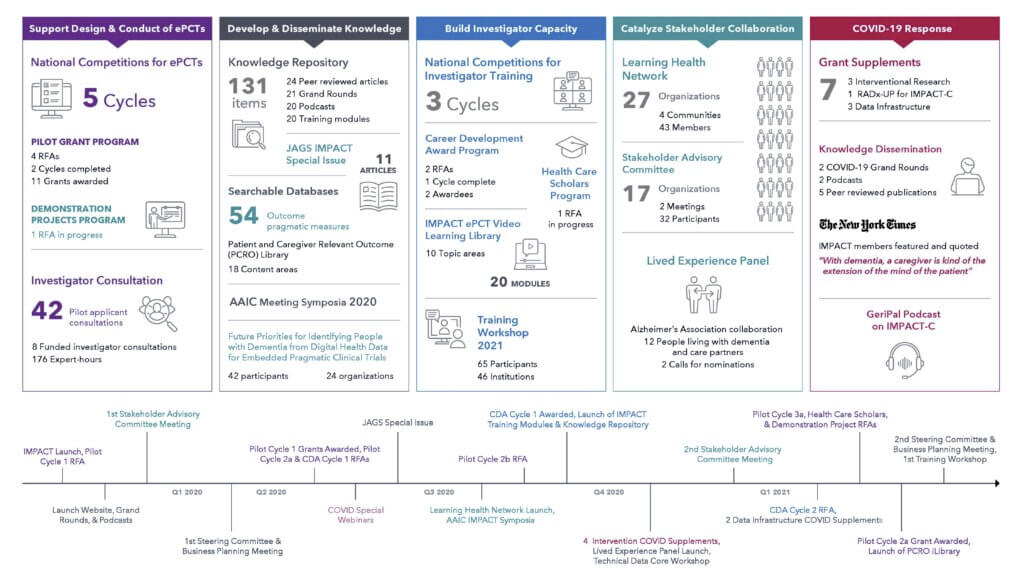
The National Institute on Aging (NIA) Imbedded Pragmatic Alzheimer’s Disease (AD) and AD-Related Dementias (AD/ADRD) Clinical Trials (IMPACT) Collaboratory has been awarded two COVID-19 Supplements partnering with major retail pharmacies to create a massive monitoring system of the long-term safety and efficacy of COVID-19 vaccination on people with AD/ADRD.
The monitoring system was first established in February and is led by IMPACT Multiple Principal Investigator Vincent Mor, PhD. Mor and his team initially created the monitoring system through a partnership with Acumen, LLC and CVS, with data from more than 13 million CVS customers. The newest supplement, announced May 10 2021, adds data from 13.2 million Walgreens customers, effectively doubling the size of the securely monitored data pool. The combined system matches pharmacy customer records from CVS and Walgreens with Medicare claims to create a data infrastructure that is updated weekly, which will provide near real-time insight into the use, effects and outcomes related to use of COVID vaccines among this frail population and introduce the potential of using big data to improve our national response to the pandemic and public health outcomes for older adults.
“Since society cannot wait to vaccinate this population with an effective vaccine, we must establish an active post-vaccination surveillance system of a large population to report vaccine safety,” Mor said. “Monitoring rare adverse events requires very large populations. This project is allowing us to determine the immediate and long-term effects of vaccination on tens of millions of elderly people — nearly half the population of Medicare beneficiaries.”
These two studies are:
Monitoring Medicare Beneficiaries’ Response to COVID vaccines (CVS Project)
This project established an active post-vaccination surveillance system of a large population to track and report safety and outcomes related to SARS-CoV-2 vaccine(s). Working jointly with CVSHealth and the Medicare & Medicaid Resource Information Center (MedRIC) at Acumen, LLC, the project established a data infrastructure to monitor the effects of COVID-19 vaccines administered to Medicare beneficiaries by CVS across the country. Data transfers from the national retail pharmacy chain are merged with Medicare data. The resulting data base is analyzed to identify demographic, race/ethnicity, clinical (e.g., ADRD), and health care system factors associated with being vaccinated, estimate the rate of adverse events attributable to the vaccine and estimate breakthrough COVID illness among vaccinated Medicare beneficiaries. This program provides the basis for assessing mid- to long-term safety and efficacy of the different vaccines being distributed for use in the elderly population in the country.
Medicare Match for post-vaccination monitoring (Walgreens)
This project builds on the researchers’ partnership with CVS to establish a data infrastructure to monitor the effects of COVID-19 vaccines administered to Medicare beneficiaries by CVS across the country. The goal of this project is to add data from Walgreen’s 13,231,175 million Medicare beneficiary pharmacy customers to the existing infrastructure which will increase the research team’s capacity to track immediate and long term effects of vaccination on nearly half the population of Medicare beneficiaries.
The need for a larger study population is particularly important because those with ADRD already suffering from neurodegenerative diseases may be at greater risk of serious neurological adverse events of vaccines. In addition, the unknown potential for adverse events may result in high rates of incomplete vaccination (both shots) among older adults. Such uncertainty contributes to vaccine hesitancy and may result in large disparities in SARS-CoV-2 vaccination among racial/ethnic groups, for which disparities in seasonal influenza vaccination already exist.
To learn more about these studies, visit this page on Building Infrastructure Supplement Awards, and the press release from Brown University at this link.