The NIH Pragmatic Trials Collaboratory aims to improve the way clinical trials are conducted by creating a new infrastructure for collaborative research with healthcare systems, ultimately ensuring that healthcare providers and patients can make decisions based on the best available clinical evidence. The Collaboratory supports the design and rapid execution of pragmatic clinical trial Demonstration Projects to address questions of major public health importance and engage healthcare delivery systems in research partnerships. The Collaboratory also provides training resources on how to design, conduct, and disseminate embedded pragmatic clinical trials (ePCTs).
Ethics and Regulation
Public Attitudes toward Consent When Research Is Integrated into Care—Any “Ought” from All the “Is”?
Hey and Taljaard co-authors on review of pragmatic clinical trials
March 28, 2021
IMPACT Collaboratory members Spencer Hey, PhD and Monica Taljaard, PhD are among the authors of a review of pragmatic clinical trials recently published online in the Journal of Clinical Epidemiology and covered in a blog post from the US GRADE network. The first author of the study is Stuart G. Nicholls, PhD, of the Ottawa Hospital Research Institute.
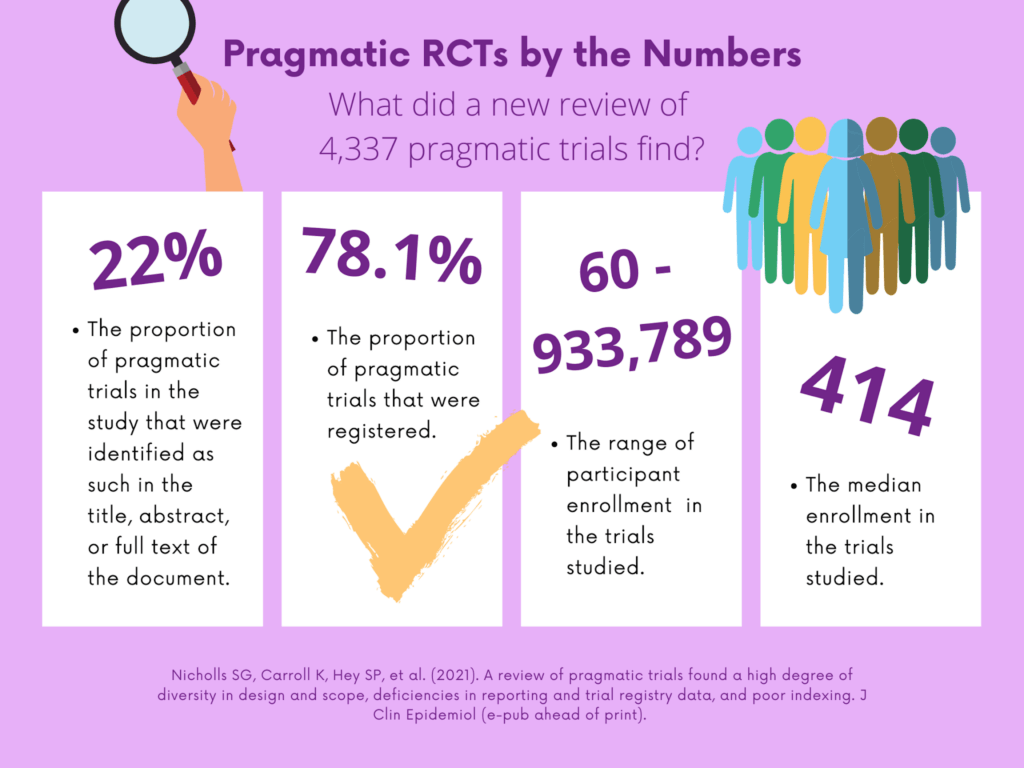
The researchers identified 4,337 eligible trials in their review of data collected from MEDLINE, Web of Science, ClinicalTrials.gov and full text between January 2014 and April 2019. They found diversity in research areas, but only 22% of the trials identified were labeled as pragmatic in the title, abstract or full text of the document. In addition, the authors estimated one-fifth of trials under-accrued by at least 15%. They concluded that there is a need to improve reporting of pragmatic trials and quality of trial registry data. Under accrual remains a challenge in pragmatic RCTs despite calls for more streamlined recruitment approaches.
The US GRADE Network blog developed an infographic to share the findings of the review.
Podcast 14: Ethical challenges with pragmatic RCTs: General issues and special considerations in dementia
 Stuart Nicholls, PhD, from the Clinical Epidemiology Program at the Ottawa Hospital Research Institute, continues the discussion from his February 18 Grand Rounds presentation. This discussion with IMPACT Principal Investigator Susan Mitchell, MD, MPH answers questions from Dr. Nicholls's presentation on giving an overview of ethical issues raised by pragmatic randomized controlled trials (RCTs) and which derive from ongoing work to develop guidance for those designing and conducting pragmatic RCTs.
Stuart Nicholls, PhD, from the Clinical Epidemiology Program at the Ottawa Hospital Research Institute, continues the discussion from his February 18 Grand Rounds presentation. This discussion with IMPACT Principal Investigator Susan Mitchell, MD, MPH answers questions from Dr. Nicholls's presentation on giving an overview of ethical issues raised by pragmatic randomized controlled trials (RCTs) and which derive from ongoing work to develop guidance for those designing and conducting pragmatic RCTs.
Want to hear more? View the full Grand Rounds presentation.
For alerts about new episodes, subscribe free on iTunes or SoundCloud.